Occurrence of pharmaceuticals and risk assessment in urban groundwater
Francesc Labad
Laura Scheiber
Rotman Criollo
Olha Nikolenko
Sandra Pérez
Antoni Ginebreda
Due to the fast urbanization and climate change, urban aquifers are considered as a strategic source of potable water. However, a potential limitation is the presence of contaminants, such as pharmaceuticals, which might deteriorate groundwater quality. This work investigated the occurrence of pharmaceuticals and evaluated their human health risk in an alluvial urban aquifer recharged by a polluted river that receives discharges from wastewater treatment plants (WWTPs). To this end, river and groundwater samples were collected from February to May 2021 for the analysis of 92 pharmaceuticals using a solid-phase extraction and a high-pressure liquid chromatography coupled to a high resolution mass spectrometric methodology (HPLC-HRMS). Results showed that 35 pharmaceuticals, including 6 transformation products (TPs), were detected in all groundwater samples and the range of concentrations was from the low ng L−1 to 44.5 µg L−1. Moreover, the concentrations of some substances decreased along the flow path during bank filtration, suggesting the occurrence of natural attenuation processes (e.g., adsorption or oxidation-reduction). Finally, most of the measured substances did not pose a risk to human health since estimated risk quotients were low.
- Article
(2283 KB) - Full-text XML
-
Supplement
(493 KB) - BibTeX
- EndNote
Sustaining healthy living conditions in urban areas is a tremendous challenge facing the European Union, and the pivotal issue is the provision of good quality freshwater resources under the pressure of rapid urban growth and climate change. Urban aquifers might be considered as an important source for water supply, however, groundwater in urban settings is vulnerable to contamination caused by anthropogenic activities such as the leakage from sewers and the discharge of wastewaters, the increase in urbanized areas and sealed surfaces, the interaction with underground constructions and surface water bodies etc. As a result, different organic and inorganic contaminants are detected in urban aquifers (Burri et al., 2019).
Among these pollutants, pharmaceuticals have drawn attention of the scientific community due to their increasing consumption and their widespread presence in the aquatic environment (Parra-Saldivar et al., 2020). These substances belong to different therapeutic groups such as antibiotics, anti-inflammatories and analgesics, psychiatric drugs, lipid regulators and anti-hypertensives, among others. Pharmaceuticals enter urban groundwater through different pollution sources. Their main pollution source is the excretion via urine and faeces in wastewater, which might contain the original compound but also its conjugated and transformed forms (Singh, 2014). Some pharmaceuticals are not fully removed by WWTPs and might be found at the concentrations ranging from ng L−1 to µg L−1 in their effluents that are discharged into adjacent aquatic environment including subsurface water bodies (Khasawneh and Palaniandy, 2021).
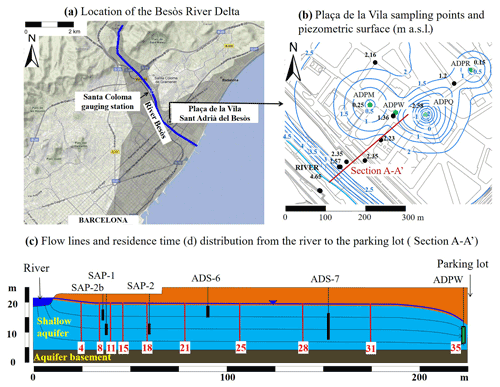
Figure 1(a) Location of the Besòs River Delta (Barcelona, NE Spain), (b) piezometric surface (in m a.s.l.) of the study area and sampling points and (c) Profile A–A' showing the major direction of the groundwater flow and the residence time (in days) indicated with red colour inside the boxes for different sections of the profile. This figure was adapted from Jurado et al. (2021).
Once in the aquifer their evolution is highly uncertain, as their behaviour depends on the coupled thermo-hydro-chemical processes that occur simultaneously in the subsurface. The first step to understand these processes is to predict the spatial and temporal distribution of the concentrations of pharmaceuticals in groundwater, which implicitly requires the information about their concentrations in the potential contamination sources, the aquifer residence time etc. (Jurado et al., 2021). Therefore, considering the increasing consumption of pharmaceuticals and the rising demand for freshwater resources in urban settings, investigating the occurrence of a wide array of pharmaceuticals in groundwater is of paramount importance to the use of groundwater as a safe drinking water source.
In this context, the objectives of this research were: (1) to investigate the occurrence of 92 pharmaceuticals, including 12 transformation products (TPs) in an urban aquifer and (2) to conduct the human health risk assessment. Groundwater samples were collected from the shallow aquifer of the Besòs River Delta (Barcelona, NE Spain) and the Besòs River, which is the main aquifer contamination source.
2.1 Study area
The study area is located in the lower part of the Besòs River Delta (Barcelona, NE Spain) (Fig. 1a). It belongs to the Mediterranean climate zone, in which January is the coldest month and July the warmest one, with an average temperature of 7.5 and 23 ∘C, respectively. Annual average precipitation is 600 mm, although precipitation varies significantly through the year.
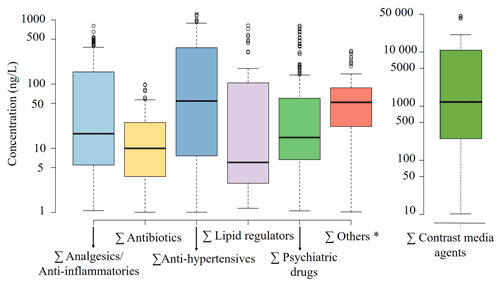
Figure 2Box-plot of the concentration of pharmaceuticals grouped according to different therapeutic groups in the urban groundwater of the shallow aquifer of the Besòs Delta River. The dots represent the outliers. * Others include the antifungals fluconazole and climbazole, the antiparasitic crotamiton and the local anaesthetic lidocaine.
Pharmaceutical substances were investigated in the unconfined shallow aquifer, which consists of sands. This aquifer is hydraulically connected to the polluted Besòs River that is the major recharge source (Fig. 1c). Consequently, this river controls the chemical characteristics of the groundwater within the shallow aquifer. However, additional sources pharmaceuticals contamination are leakage from the sewage system and urban runoff (Jurado et al., 2013). The river aquifer system is very dynamic due to a constant pumping of about 150–200 L s−1 conducted to avoid seepage problems in an underground parking lot located 200 m below the river (pumping wells ADPW, ADPM, ADPQ and ADPR from Fig. 1b). Groundwater flows from the Besòs River to the underground parking area with an average residence time of 35 d (Fig. 1c).
2.2 Sampling and analytical method
The 6 sampling points considered in this study are the pumping well W (ADPW), the piezometers of the series SAP-n (located close to the river) and ADS-n (Fig. 1b). Water samples for the analysis of pharmaceuticals were collected from February to May 2021. In total four surface water samples from the Besòs River and 24 groundwater samples were collected. The latter were obtained after stabilization of field parameters (i.e., pH and electrical conductivity) or by pumping at least three times the volume of water present in the piezometer. Samples were put in a field refrigerator and taken to the laboratory, where they were vacuum filtered through 0.7 µm glass fibre filters and stored at −20 ∘C until analysis. Finally, groundwater and river samples were analysed using a high-pressure liquid chromatography (HPLC) coupled to a high-resolution mass spectrometer (HRMS), the Q-Exactive Orbitrap. Initially this method was developed by Fagnani et al. (2022) for the analysis of surface water samples but was adapted for to the study of groundwater samples. This technique is characterized by a high sensitivity with detection limits between 0.03 and 0.32 ng L−1.
2.3 Evaluation of human health risk
Maximum concentrations of the ubiquitous pharmaceuticals (detection frequencies of 100 %) in groundwater were used to estimate the risk quotients (RQs) for human health to foresee the “worst-case” scenario. The RQs were evaluated as follows:
where Cmax is the maximum concentration of a given substance in groundwater and DWEL is the drinking water equivalent level, which was computed using Eq. (2) (de Jesus Gaffney et al., 2015):
where ADI is the Acceptable Daily Intake (µg kg−1 d), BW is the body weight (kg) and DWI is the water daily ingestion rate (L d−1), which were assumed to be 65 kg and 2.25 L d−1 (EFSA, 2010), respectively. HQ represents the hazard quotient assumed to be 1, AB is the gastrointestinal absorption and the value considered was 1 (i.e., adsorption rate is 100 %), and FOE is the frequency of exposure assumed to be 1 (i.e., 365 d of the year). ADI values were selected according to previous investigations (Table 1), ranging from 0.057 µg kg−1 d for the antihypertensive valsartan to 125 000 µg kg−1 d for the contrast agent iohexol (Liu et al., 2019; Parida et al., 2021) (Table 1).
The risk criteria applied for the evaluated RQs were the following: RQs lower than 0.1 mean no risk, RQs between 0.1 and 1 indicate median risk, and RQs higher than 1 potentially affect human health (Martín et al., 2011).
Table 1ADI values, maximum (Max.) concentration DWEL values and RQmax for the target pharmaceuticals. Metabolites are highlighted in bold.
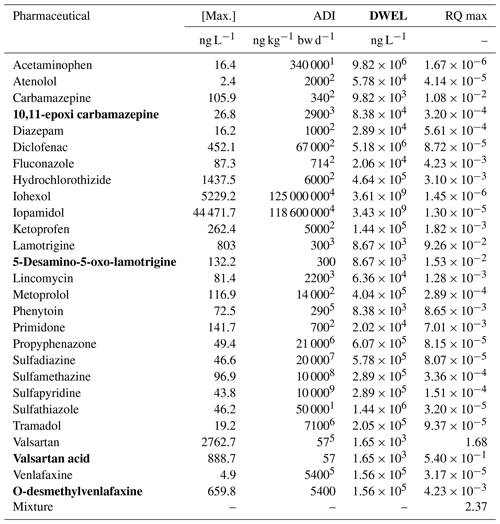
References: 1 Schwab et al. (2005), 2 Sengar and Vijayanandan (2022), 3 Prosser and Sibley (2015), 4 Parida et al. (2021), 5 Liu et al. (2019), 6 de Jongh et al. (2012), 7 OCS (2022), 8 Zhu et al. (2018) and 9 NRA (2000).
3.1 General results
Results showed that 35 pharmaceuticals were ubiquitous in groundwater belonging to the following therapeutic groups: 14 psychiatric drugs, 6 anti-hypertensives, 5 analgesics/anti-inflammatories, 5 antibiotics, 1 contrast media agent, 1 lipid regulator, 1 antifungal, 1 local anaesthetic and 1 antiparasitic. The ranges of concentrations varied from concentrations below the limit of quantification, which usually are 0.30 ng L−1, to 44.5 µg L−1 for the contrast media agent iopamidol (Table S1 in the Supplement). The therapeutic groups that most contribute to the groundwater contamination were contrast media agents followed by anti-hypetensives (Fig. 2). In contrast, the group of lipid regulators demonstrated the lowest median concentration (Fig. 2). There were 7 substances with average concentrations exceeding the threshold of 300 ng L−1, namely in descending order: 12.7 µg L−1 for iopamidol, 762.5 ng L−1 for valsartan, 647.7 ng L−1 for iohexol, 456.5 ng L−1 for hydrochlorothiazide, 397.2 ng L−1 for lamotrigine, 366.8 ng L−1 for the TP valsartan acid and 301.4 ng L−1 for furosemide (Table S1). In general, most pharmaceuticals showed lower concentrations in groundwater than in the Besòs River, suggesting that some natural attenuation mechanisms such as redox or adsorption processes might have occurred when river water infiltrated the aquifer.
3.2 Temporal evolution
Figure 3 shows the total concentration of pharmaceuticals in the river and the groundwater sampling points for the different campaigns. The maximum river concentration (67.4 µg L−1) occurred in February and the minimum (25.6 µg L−1) was in April. In February and March surface water samples demonstrated higher concentrations of pharmaceuticals in comparison to their concentrations in groundwater. However, the total concentrations of pharmaceuticals at the shallowest points SAP-2b and ADS-6 were higher than those of the river in April and May 2021 (Fig. 3). It suggests that a composite river water sample should be collected, as the concentration of target pharmaceuticals might vary during the day depending on the time that effluents from WWTPs are discharged into the river.
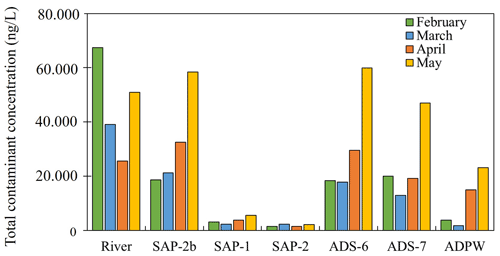
Figure 3Total contaminant concentration in Besòs River and groundwater from February to May 2021 (C1-February, C2-March, C3-April and C4-May).
Concerning groundwater samples, the aforementioned shallow points SAP-2b and ADS-6 showed the highest concentrations of pharmaceuticals, especially in May 2021. On the contrary, the deepest points SAP-1 and SAP-2 showed the lowest concentrations of pharmaceuticals throughout the four months. Considering a flow line from SAP-2b to the parking area, it seems that the total contamination of pharmaceuticals decreased from SAP-2b to SAP-2 in all the sampling campaigns. At the same time, the concentration increased at the observation points located outside the riparian zone of the river within the paved area (ADS-6, ADS-7 and ADPW, Fig. 1b) (Fig. 3), suggesting the presence of additional sources of contamination, such as the infiltration of the urban runoff or the leakage from the sewerage network (Jurado et al., 2013).
Some pharmaceuticals might be used as potential groundwater tracers as their average concentrations during the four sampling campaigns exceeded the threshold of 100 ng L−1, suggesting their persistent nature. That was the case of lamotrigine, furosemide, hydrochlorothiazide, valsartan and its TP valsartan acid, iopamidol, gemfibrozil, gabapentin, the TP of venlafaxine O-desmethylvenlafaxine and lidocaine. TPs were usually detected at lower concentrations than their parent substances (e.g., 5-desamino-5-oxo-lamotrigine, 10,11-epoxi carbamazepine or 4-hydroxy diclofenac, Table S1). However, there was one exception for the TP O-desmethylvenlafaxine, which average concentrations were two orders of magnitude larger than its parent compound (2.1 vs. 200.2 ng L−1 in February, 0.92 vs. 158.5 ng L−1 in March, 0.3 vs. 203.7 ng L−1 in April and 0.9 vs. 303.9 in May 2021).
Human health risk was evaluated for pharmaceuticals that were detected in all groundwater samples and for non-ubiquitous substances whose average concentration were higher than 100 ng L−1. Additionally, the TPs valsartan acid, O-desmethylvenlafaxine and 5-Desamino-5-oxo-lamotrigine were included into this evaluation using the ADIs of their parent compounds, as their maximum concentrations in groundwater were high (888.7, 659.8 and 132.3 ng L−1, respectively).
The threat that pharmaceuticals pose to human beings was assessed through RQ comparing the maximum concentration of individual pharmaceutical in groundwater to the DWEL values. Results of DWEL values and RQMAX are summarized in Table 1. Generally, DWEL values were frequently several orders of magnitude higher than the maximum concentration of pharmaceuticals in groundwater (Table 1). As a result, most RQs were far below 0.01, indicating that these substances did not pose a risk for human health in case that groundwater was used as drinking water. There were only two exceptions, valsartan and its TP valsartan acid that showed RQ values of 1.68 and 0.54, respectively. Thus, these pharmaceuticals might potentially affect human health or represent a moderate risk. Moreover, the risk assessment of individual pharmaceuticals, are likely to be underestimated as these substances were detected simultaneously in groundwater samples. For this reason, the RQ of the mixture of these substances was evaluated by summing individual RQs. The mixture might represent a potential risk for human health as its RQ value is 2.37 (Table 1).
Water shortage triggers the exploration of alternative water resources such as urban groundwater. However, the continuous inflow of pharmaceutical substances into the environment can limit the potential use of urban aquifers for potable water supply, as it might pose a threat to human health. In this context, the described above study monitored the occurrence of a several pharmaceuticals and their TPs in an urban aquifer, which were detected at a wide range of concentrations, reaching up to 44.5 µg L−1. Results showed that the average concentrations of pharmaceuticals in river water were higher than those found in the aquifer, suggesting that natural attenuation processes take place during the infiltration of surface water into the subsurface. However, some pharmaceuticals were detected at average concentrations higher than 100 ng L−1, which might indicate that they are suitable tracers to track anthropogenic sources of groundwater contamination. Finally, most of the studied substances did not represent a threat for human health as estimated risk quotients were below 0.1.
Data on the concentrations of pharmaceuticals can be send under reasonable request to the corresponding author.
The supplement related to this article is available online at: https://doi.org/10.5194/adgeo-59-1-2022-supplement.
AJ has contributed to the Conceptualization, Methodology, Data, Curation; Writing Original Draft, Visualization and Funding acquisition. FL has participated in the Methodology and Data Curation. LS and ON have contributed to the Methodology and Data curation. RC has prepared part of the Methodology and Visualization. SP has contributed in the Methodology and Resources. AG has participated in the Conceptualization, Writing Original Draft and Funding acquisition. All authors have contributed to manuscript reviewing and editing.
The contact author has declared that none of the authors has any competing interests.
Publisher’s note: Copernicus Publications remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the special issue “Quality and quantity issues in urban hydrogeology (EGU2022 HS8.2.8 session)”. It is a result of the EGU General Assembly 2022, Vienna, Austria, 23–27 May 2022.
This research has been supported by the Ministerio de Ciencia e Innovación (grant no. CEX2018-000794-S).
We acknowledge support of the publication fee by the CSIC Open Access Publication Support Initiative through its Unit of Information Resources for Research (URICI).
This paper was edited by Victor Vilarrasa and reviewed by Pablo Gamazo and two anonymous referees.
Burri, N. M., Weatherl, R., Moeck, C., and Schirmer, M.: A review of threats to groundwater quality in the anthropocene, Sci. Total Environ., 684, 136–154, https://doi.org/10.1016/j.scitotenv.2019.05.236, 2019.
de Jesus Gaffney, V., Almeida, C. M. M., Rodrigues, A., Ferreira, E., Benoliel, M. J., and Cardoso, V. V.: Occurrence of pharmaceuticals in a water supply system and related human health risk assessment, Water Res., 72, 199–208, https://doi.org/10.1016/j.watres.2014.10.027, 2015.
de Jongh, C. M., Kooij, P. J. F., de Voogt, P., and ter Laak, T. L.: Screening and human health risk assessment of pharmaceuticals and their transformation products in Dutch surface waters and drinking water, Sci. Total Environ., 427–428, 70–77, https://doi.org/10.1016/j.scitotenv.2012.04.010, 2012.
EFSA, N.: Scientific Opinion on Dietary Reference Values for Water, EFSA Journal 2010, 8, 1459.
Fagnani, E., Montemurro, N., and Pérez, S.: Multilayered solid phase extraction and ultra performance liquid chromatographic method for suspect screening of halogenated pharmaceuticals and photo-transformation products in freshwater – comparison between data-dependent and data-independent acquisition mass spectrometry, J. Chromatogr. A, 1663, 462760, https://doi.org/10.1016/j.chroma.2021.462760, 2022.
Jurado, A., Vàzquez-Suñé, E., Soler, A., Tubau, I., Carrera, J., Pujades, E., and Anson, I.: Application of multi-isotope data (O, D, C and S) to quantify redox processes in urban groundwater, Appl. Geochem., 34, 114–125, https://doi.org/10.1016/j.apgeochem.2013.02.018, 2013.
Jurado, A., Vázquez-Suñé, E., and Pujades, E.: Urban Groundwater Contamination by Non-Steroidal Anti-Inflammatory Drugs, Water, 13, 720, https://doi.org/10.3390/w13050720, 2021.
Khasawneh, O. F. S. and Palaniandy, P.: Occurrence and removal of pharmaceuticals in wastewater treatment plants, Process Safe. Environ. Protect., 150, 532–556, https://doi.org/10.1016/j.psep.2021.04.045, 2021.
Liu, M., Yin, H., and Wu, Q.: Occurrence and health risk assessment of pharmaceutical and personal care products (PPCPs) in tap water of Shanghai, Ecotoxicol. Environ. Safe., 183, 109497, https://doi.org/10.1016/j.ecoenv.2019.109497, 2019.
Martín, J., Camacho-Muñoz, D., Santos, J. L., Aparicio, I., and Alonso, E.: Monitoring of pharmaceutically active compounds on the Guadalquivir River basin (Spain): occurrence and risk assessment, J. Environ. Monit., 13, 2042, https://doi.org/10.1039/c1em10185d, 2011.
NRA: National Registration Authority for Agricultural and Veterinary Chemicals (2000), The NRA Review of sulphonamides. Final Report. August 2000 NRA Review Series 00.3, National Registration Authority for Agricultural Chemicals, Canberra, Australia, https://apvma.gov.au/sites/default/files/publication/15001-sulphonamides-review-final-report.pdf (last access: 1 February 2022), 2000.
OCS: Office of Chemical Safety, Department of Health, Australian Government, ADI List: Acceptance Daily Intakes for Agriculture and Veterinary Chemicals, Commonwealth Copyright, Australia, ISSN 1446-1412 (electronic), 100 pp., 2022.
Parida, V. K., Saidulu, D., Majumder, A., Srivastava, A., Gupta, B., and Gupta, A. K.: Emerging contaminants in wastewater: A critical review on occurrence, existing legislations, risk assessment, and sustainable treatment alternatives, J. Environ. Chem. Eng., 9, 105966, https://doi.org/10.1016/j.jece.2021.105966, 2021.
Parra-Saldivar, R., Castillo-Zacarías, C., Bilal, M., Iqbal, H. M. N., and Barceló, D.: Sources of Pharmaceuticals in Water, Springer Berlin Heidelberg, Berlin, Heidelberg, https://doi.org/10.1007/698_2020_623, 2020.
Prosser, R. S. and Sibley, P. K.: Human health risk assessment of pharmaceuticals and personal care products in plant tissue due to biosolids and manure amendments, and wastewater irrigation, Environ. Int., 75, 223–233, https://doi.org/10.1016/j.envint.2014.11.020, 2015.
Schwab, B. W., Hayes, E. P., Fiori, J. M., Mastrocco, F. J., Roden, N. M., Cragin, D., Meyerhoff, R. D., D'Aco, V. J., and Anderson, P. D.: Human pharmaceuticals in US surface waters: A human health risk assessment, Regulat. Toxicol. Pharmacol., 42, 296–312, https://doi.org/10.1016/j.yrtph.2005.05.005, 2005.
Sengar, A. and Vijayanandan, A.: Human health and ecological risk assessment of 98 pharmaceuticals and personal care products (PPCPs) detected in Indian surface and wastewaters, Sci. Total Environ., 807, 150677, https://doi.org/10.1016/j.scitotenv.2021.150677, 2022.
Singh, A.: Groundwater resources management through the applications of simulation modeling: A review, Sci. Total Environ., 499, 414–423, https://doi.org/10.1016/j.scitotenv.2014.05.048, 2014.
Zhu, M., Zhao, H., Chen, J., Xie, H., and Du, J.: Investigation of antibiotics in sea cucumbers: occurrence, pollution characteristics, and human risk assessment, Environ. Sci. Pollut. Res., 25, 32081–32087, https://doi.org/10.1007/s11356-018-3131-7, 2018.
- Abstract
- Introduction
- Materials and methods
- Occurrence of pharmaceuticals in urban groundwater and river water
- Human health risk assessment
- Conclusions
- Data availability
- Author contributions
- Competing interests
- Disclaimer
- Special issue statement
- Financial support
- Review statement
- References
- Supplement
- Abstract
- Introduction
- Materials and methods
- Occurrence of pharmaceuticals in urban groundwater and river water
- Human health risk assessment
- Conclusions
- Data availability
- Author contributions
- Competing interests
- Disclaimer
- Special issue statement
- Financial support
- Review statement
- References
- Supplement




